Navigation : EXPO21XX > RENEWABLE ENERGY 21XX >
H33: Universities - Renewable Energy Research
> ASU Center for Bio-Inspired Solar Fuel
ASU Center for Bio-Inspired Solar Fuel

Videos
Loading the player ...
- Offer Profile
- Our objective is to adapt
the fundamental principles of natural photosynthesis to the man-made
production of hydrogen or other fuels from sunlight
A multidisciplinary team will research artificial photosynthetic antennas and reaction centers that absorb light efficiently and convert it to electrochemical energy, a water oxidation catalyst based on that found in photosynthesis and assembled in a way that mimics the process used by nature,and an electron accumulator and proton reduction catalyst based on natural hydrogenase enzymes. The antennas and reaction centers will be designed using the techniques of organic chemistry. The catalysts will be developed using peptide engineering methods. These components will be structurally organized using concepts from materials science, nanotechnology and nucleic acid engineering.
Product Portfolio
Mission
- Our mission is to construct a complete system for
solar-powered production of fuels such as hydrogen via water splitting.
Design principles will be drawn from the fundamental concepts that underlie
photosynthetic energy conversion.
A major challenge facing humanity is developing a renewable source of energy to replace our reliance on fossil fuels. The ideal source will be abundant, inexpensive, environmentally clean, and widely distributed geographically. Energy from the sun meets these criteria. Unfortunately, practical, cost effective technologies for conversion of sunlight directly into useful fuels do not exist, and new basic science is required. A blueprint for storage of solar energy in fuels does exist, however, in photosynthesis. Indeed, all of the fossil-fuel-based energy we consume today derives from sunlight that was harvested by photosynthetic organisms.
Recognizing the need for new science, the DOE established the ASU Center for Bio-Inspired Solar Fuel Production in 2009. The Center involves 11 faculty from the Department of Chemistry and Biochemistry and is housed within the ASU Center for Bioenergy and Photosynthesis. The Solar Fuel Center consists of faculty, research associates, graduate students and undergraduate researchers dedicated to solving the renewable energy problem.
Center Objective
Our objective is to adapt the fundamental principles of natural photosynthesis to the man-made production of hydrogen or other fuels from sunlight
A multidisciplinary team will research artificial photosynthetic antennas and reaction centers that absorb light efficiently and convert it to electrochemical energy, a water oxidation catalyst based on that found in photosynthesis and assembled in a way that mimics the process used by nature,and an electron accumulator and proton reduction catalyst based on natural hydrogenase enzymes. The antennas and reaction centers will be designed using the techniques of organic chemistry. The catalysts will be developed using peptide engineering methods. These components will be structurally organized using concepts from materials science, nanotechnology and nucleic acid engineering. The Science
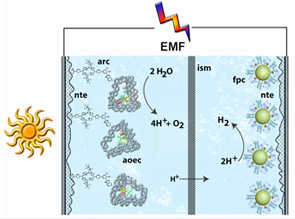
- Water oxidation complexes will be based on a
unique, self-assembling, engineered DNA nanostructure that organizes short
synthetic peptides arranged in a manner analogous to the natural
oxygen-evolving complex. These peptides will be used to construct a
metal-ion-based catalytic site similar to the natural one, using assembly
methods found in photosynthesis. In a second approach, peptide-based
water-soluble analogs of the natural photosynthetic oxygen-evolving complex
will be sought. The DOE ALS in Berkeley will be used for X-ray (as
necessary), XAFS and XANES characterization of the artificial water
oxidation (and proton reduction) catalysts.
Hydrogen production catalysts will be based on natural hydrogenase enzymes. Iron-containing catalytic sites and iron-sulfur sites for storing reduction equivalents will be organized into functional catalysts using metal nanoparticles and linked to transparent electrodes. New transparent, nanostructured, high-surface-area conducting metal oxide materials will be constructed to serve as functional frameworks for organizing the various components of the system, separating mutually reactive intermediates, and facilitating electrical communication among components.
A major challenge is the integration of the various components mentioned above into a functional system that is competent to carry out water splitting as a unit. This will require careful attention both to the thermodynamic and kinetic properties of the catalysts and charge-separation units and to the transport of redox equivalents and materials among the various units of the complex. Thus, the research has a strong systems engineering component. Two photosystems, à la photosynthesis, will likely be necessary to achieve useful efficiencies. Initially, metallic connections between some subsystems will be used in order to permit testing of components electrochemically and application of external emf as necessary. Based on the performance of natural photosynthesis, the synthetic system has the potential to produce fuel efficiently from sunlight and water, to be inexpensive, to use earth-abundant elements, and to be a practical solution to humanity’s energy problems. Realizing this potential is a significant fundamental and applied scientific challenge.
While pursuing this ambitious goal, the Center will uncover basic scientific knowledge that will point the way to new catalysts for water splitting and fuel cells, new materials for solar photovoltaics of various kinds, new ways to use DNA and peptides for preparation of artificial enzymes for biomedical and other technological applications, and new fundamental ways of understanding and manipulating matter that will have applications in many different areas of technology. It may also help identify ways to modify natural photosynthesis in plants so that it can better fill humanity’s needs.